Introduction
This cover the design, process, testing and putting into the operation of an Incinerator, together with supporting document of the equipment to be used in a pharmaceutical solid waste burning. This design must meet all cGMP, GAMP, ISO 14001 & OSHAS 18001 guidelines and legislation.
2.1 Purpose
This document was generated under the authority of Orion Pharma Ltd. for the purpose of specify the user requirement for an Incinerator. The User Requirement Specification (URS) is provided to to procure a functional Incinerator that meet the user’s needs in the most cost-effective method possible. The URS is then provided to the supplier to provide a price quote for purchase of the Incinerator including the design and manufacturing of the equipment.
2.2 Scope
This user requirement specification will be recognized as an integral part of the procurement agreement with the selected equipment vendor. The equipment supplier or vendor will be abide by the information and conditions set forth by this document as well as the standard purchasing term and conditions of Orion Pharma Ltd.
2.3 General Description
The Incinerator will be used for burning tablet, Aluminium foil, pvc foil, paper, carton etc
2.4 Validation Plan
The machine will undergo the qualification and validation activities stated below. All qualification and validation protocols and reports, according to the subchapters below, need to be formally approved by QA, Production & Engineering. The equipment supplier or vendor has specific responsibilities as outlined by the Validation Plant and this URS.


![]()
![]()


![]()
3 Overview
3.1.1 Procedure
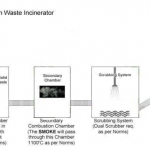
· Destroy the wasted materials or chemical uses in pharmaceuticals through Primary & Secondary combustion chambers with working temperatures are 850° C & 1100° C or 1200° C & 1400° C. The incinerator design must meet with ISO, OSHAS, Emission Standards, Norms and Guidelines. The incinerator design must have heat recovery system and low fuel consumption system while incineration.
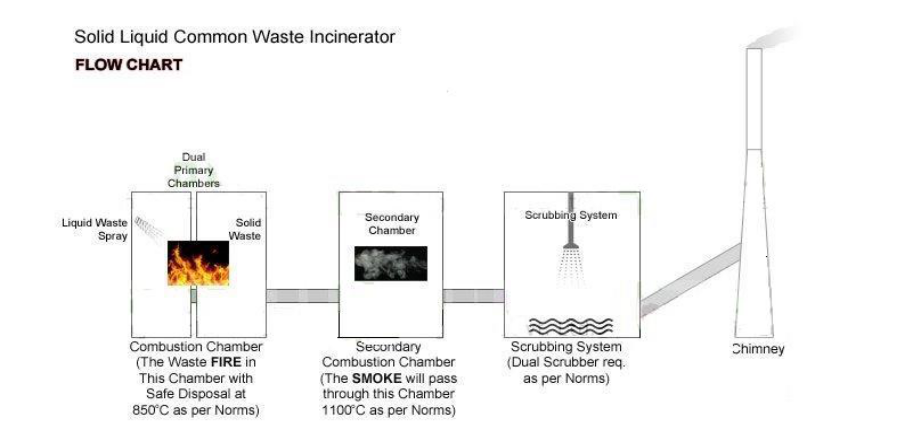
3.1.2 Capacity
· Capacity: 300 KG.
3.1.3 Output Air Quality:
· Output air quality must meet with local regulatory, ISO, OSHAS, IS Emission standard.
Article 4: Specifications
4.1 GENERAL
· Waste Incinerator system with complete setup designed for pharmaceuticals industry.
Waste Type: Solid (Tablet, Powder etc.) Aluminium, PVC & PVDC Foil
Paper, others
4.2 Function
The machine should have following features in its respective parts –
· Feeding system
· Automatic/ and manual control system
· Automatic heating control system
· Easy maintenance
· Outdoor type IP grade material
· Heat recovery system
· Low fuel consumption system
· Combustion chamber dual type with heat recovery system
· Burner system with economizer / low fuel method
· Scrubbing system as per regulation
· Easy dust collecting system
· Chimney (height 50 meter)
· Record printing system
4.3 Technical Data
· Power Supply : 380~415Volts AC, 50 Hz
· Fuel Type : Both Option for Diesel and Gas
· Capacity : 250 kg
· Combustion efficiency : 99.99%
· Chimney Height : Above 50 Meter
· Operating temperature : 1200⁰ C
4.6 Safety
There should be standard safety facilities-
– All regulation must be incorporated in the design according to international standards.
Operation Safety
§ The system must have operational proper safety during feeding material and operation the machine and unwanted fume.
Environment Safety
§ The system must have proper Environment safety as per ISO 14001 & OSHAS 18001 guideline.
Emergency Stop
§ The system must have a local stop capability that shuts the system down in an orderly manner or safe way.
§ The E-stop mechanism shall be located in easily accessible areas around the equipment. No damage to the machine will occur due to emergency stop.
§ All operators setting shall be unchanged and restored after an emergency stop.
§ The unit shall not be allowed to restart without operation intervention. It shall be necessary to restore the emergency stop button the original state and reset the system before restarting.
4.7 Maintenance and Spare Parts
Maintenance
|
§ The machine shall be built with intention for quick and easy access for service and maintenance. |
|
§ The supplier shall provide Orion’s personnel an adequate training for operation and maintenance of the control system. |
|
§ The supplier shall provide lubrication list and recommended schedule |
|
§ The supplier shall provide a scheduled maintenance plan. |
Spare parts
|
§ Spare parts, recommended for two years of normal operation. (List must be attached with the offer) |
|
§ Special tools necessary for service and maintenance shall be stated as a separate post in the quotation and included in delivery. |
|
§ The supplier shall provide a list of recommended spare parts of the control system. |
ARTICLE 5: PLANS & DOCUMENTS
§ Supplier will prepare the DQ (Design qualification) and get approval from authorized representative of the client in advance.
§ The supplier shall provide the following documents, at the time of the offer submission and inspection visit/acceptance trials, all in English language.
– Equipment completeness and compliance to the specifications and design
– Process Flow Chart
– p&I Diagram
– Equipment operational compliance
– Proper IQ, OQ, protocol and report.
– All Mechanical Plans “As built Drawings”
– All Electrical Plans “As built Drawings” and SLD.
– Instruction Layout-equipments, pipes, cable trays, tanks, valves etc.
§ The following manuals / drawings to be provided by vender – (Soft & Hard Copies)
Installation Manual
2. 2. Operational Manual
3. Service Manual (Including trouble shooting and maintenace)
4. Routine maintenance schedule and change parts
5. PLC ladder diagram
6. Wiring diagram
7. Equipment layout diagram
8. Control Panel Board Electrical Assembly drawing & others necessary.
9. Complete spare lists.
10. Operator interface instruction manual with screen printed Flow chart.
Certificates
|
§ Noise levels certificate/measuring protocol. |
|
§ Calibration certificates with validity date & signature of measuring apparatus (like – pressure sensors / transmitters, Temperature sensors / transmitters, |
|
§ Training certificate. |
ARTICLE 6: RESPONSIBILITIES
6.1Supplier’s Responsibilities
· Supplier is fully responsible for supply, installation and commissioning of the system.
· Vender / supplier must follow the ISO 14001 & OSHAS 18001 guideline.
· Supplier will prepare the DQ and get approval from Orion Pharma in an advance.
· All materials of construction must comply with GMP, ISO & OSHAS requirements.
· All certificates should be handed over to Orion Pharma
· The acceptance / signature of Orion Pharma on any document provided by vender/ supplier shall not withdraw the responsibility of vender/supplier regarding the achievement of the performance criteria defined here.
· The supplier guarantees that the equipment supplied shall conform to the standards of the trade, the relevant Manufacturing Standard including Safety Standards, and the requirements of legislative or regulatory provisions applicable.
6.2 Definition of Orion Pharma’s Responsibilities
· Custom Clearance
· Transport of equipment from Chittagong to factory site at Siddhirganj, Narayanganj.
· Unloading, temporary storage, movement into position.
· Provision of suitably qualified maintenance personnel for attendance at FAT and training by supplier, both at supplier’s work on site.
· Provision of suitably qualified and experience tradesmen to conduct installation under supplier’s supervision.
· Civil work, associated with installation.
· Arrangement to make available necessary utilities (electricity, compressed air, steam, chilled water, potable water)
ARTICLE 8: SITE ACCEPTANCE TEST (SAT), COMMISSIONING,
VALIDATION AND TRAINING OF OWNER’S PERSONNEL
The supplier shall quote separately for the provision of competent English speaking, Commissioning Engineer (or Engineers) on site to:-
§ Supervise the installation of the equipment.
§ Check equipment installation, perform the start-up of the equipment and the commissioning to agreed commissioning method statements including the calibration of critical instruments, gauges etc.
Supervise Site Acceptance Tests (SAT) at site and rectify any problems.
(The supplier will be asked to provide examples of commissioning documentation at the Technical/Commercial Meeting prior to order placement).
§ The training of client’s maintenance personnel:
§ The supplier should comply with the following validation requirements as appropriate:
– Demonstrate compliance with the Specification.
– Visual inspection of the systems
– Verification of the performance data of the individual system.
– All DQ, IQ, OQ documents has to comply the cGMP standards.
– Provide drawings for Design Qualification at agreed dates.
– Record all changes and advise the client of any changes made.
– Allow inspection during construction and testing (if required by Orion Pharma).
– Advise the client of their test procedures.
– Ensure the item operates to specification.
– Provide documented proof of testing.
– .
ARTICLE-10: DOCUMENTATION STANDARDS
§ All certificates must be dated and signed.
§ Calibration certificates for testing instruments shall show the period of validity.
§ All checklists must have a signature and date by each item. (A tick or cross is not sufficient).
§ Acceptable limits must be indicated for each design parameter.
§ Full explanations must follow any results that deviate from design parameters.
§ Any alterations or corrections to the documents should be initialed and dated.
§ Correction fluid should not be used. Corrections must be made by deleting with a single line and signed and dated.
§ A statement shall be incorporated into the documentation, which details the qualification and position of every signatory.
§ Drawings must be annotated to show that they have been checked.
§ The position (Job title) and affiliation (company) of all persons signing protocols must be shown.
§ All documents should be on English.
ARTICLE 11: EXPORT PACKAGING, DELIVERY FOB, INSURANCE AND PROTECTION
The supplier will be fully responsible for all arrangements and costs of transport, insurance, taxes and duties required to deliver the equipment to nearest seaport as required in performance of the Work under the order, i.e. Orion Pharma will not be responsible for any costs associated with storage due to the supplier’s failure to comply with the Schedule of Key Dates.
It will remain the supplier’s responsibility to adequately protect equipment under his supply whilst in transportation, off loading and final positioning up to and including installation and commissioning.
Email: [email protected]
|
HICLOVER Incinerator/Parts Range |
Main Feature |
|
Medical Waste Incinerators |
HICLOVER 10-500kgs/Hr.Double Combustion Chambers |
|
Single Combustion Chamber |
Small, Cheap,10-20kgs/Hr. |
|
Three Combustion Chambers(Optional) |
3 Chambers for high risk waste |
|
Pet Cremation Equipment |
For Pet(small/big) Cremation Business |
|
Animal Incineration Equipment |
For other animal incineration |
|
Items/Model |
TS100(PLC) |
TS150(PLC) |
TS300(PLC) |
TS500(PLC) |
|
Burn Rate (Average) |
100 kg/hour |
150 kg/hour |
300 kg/hour |
500 kg/hour |
|
Control Mode |
PLC Auto. |
PLC Auto. |
PLC Auto. |
PLC Auto. |
|
Combustion Chamber |
1200L |
1500L |
2000L |
3000L |
|
Internal Dimensions |
120x100x100cm |
150x100x100cm |
170x120x100cm |
210x120x120cm |
|
Secondary Chamber |
600L |
750L |
1000L |
1500L |
|
Smoke Filter Chamber |
Dry Scrubber |
Dry Scrubber |
Dry Scrubber |
Dry Scrubber |
|
Feed Mode |
Manual |
Manual |
Manual |
Manual |
|
Voltage |
220V |
220V |
220V |
220V |
|
Power |
1.38Kw |
1.69Kw |
2.57Kw |
4.88Kw |
|
Diesel Oil Consumption (kg/hour) |
Ave.20.4 |
Ave.24.2 |
Ave.33 |
Ave.44 |
|
Natural Gas Consumption (m3n/hour) |
Ave.24.5 |
Ave.29 |
Ave.39.6 |
Ave.52.8 |
|
Temperature Monitor |
Yes |
Yes |
Yes |
Yes |
|
Temperature Protection |
Yes |
Yes |
Yes |
Yes |
|
Oil Tank |
200L |
300L |
500L |
500L |
|
Chimney |
10Meter |
10Meter |
14Meter |
14Meter |
|
Chimney Type |
Stainless Steel |
Stainless Steel |
Stainless Steel |
Stainless Steel |
|
1st. Chamber Temperature |
800℃–1000℃ |
800℃–1000℃ |
800℃–1000℃ |
800℃–1000℃ |
|
2nd. Chamber Temperature |
1000℃-1200℃ |
1000℃-1200℃ |
1000℃-1200℃ |
1000℃-1200℃ |
|
Residency Time |
2.0 Sec. |
2.0 Sec. |
2.0 Sec. |
2.0 Sec. |
|
Gross Weight |
6000kg |
8500kg |
11000kg |
16000kg |
|
External Dimensions |
260x150x180cm |
300x160x190cm |
400x210x300cm |
450x210x300cm |
 CE Certification Pass
CE Certification Pass
2020-10-28